Expertise
The added value of our ISO 13485-compliant eQMS in the development process
Anyone who develops medical technology products knows the reality:
Ideas alone aren't enough.
Between the initial product concept and a market-ready medical device lie regulatory requirements, documentation obligations, risk management, and audits. Development therefore does not take place solely in the laboratory or at a design service provider—but always within a regulated system.
For businesses, this means one thing above all else:
They need partners who can navigate this environment with confidence. That is precisely why we have consistently tailored our approach to be an integral part of this system.
We realized early on:
Efficient development is only possible when all stakeholders can seamlessly integrate into existing regulated processes. A design firm that operates independently of design controls, risk management, and documentation requirements creates additional work. In the worst case, it can even lead to regulatory risks.
When Product Complexity Requires Planning Certainty
Many of the companies we work with are mid-sized medtech manufacturers. They develop highly complex products, often over several years and involving many different disciplines. In such projects, an external development partner is not merely a source of creative inspiration. They must be able to integrate into existing processes—technically, organizationally, and regulatory. This is precisely where friction often arises.
When external partners work differently than internal development teams, additional points of interaction arise:
Documentation must be updated retroactively, decisions are difficult to trace, or processes do not align.
Our approach was therefore clear from the start:
We wanted to work in a way that would allow us to become an integral part of our clients’ development ecosystem without any friction.
That is why our design processes are modeled after the structures of regulated development projects.
Our QMS offers the following to mid-sized medtech manufacturers:
- Greater planning certainty for complex development projects - predictable costs
- Decisions are documented in a transparent manner -> no unnecessary rework
- Development steps are transparent and predictable
- Faster and more actionable decisions
- Seamless integration into existing systems
- Design processes can be categorized under design controls
- Clear interfaces with Quality Assurance (QA) and Regulatory Affairs (RA)
- Access to years of experience and an extensive network of experts
For our customers, this means one thing above all else:
planning certainty.
When it comes to complex products, it is crucial that development partners not only work creatively but also think systematically. Added to this is a factor that is often underestimated: experience. We have been working in medical technology product development for nearly 30 years. Over time, we have built up a large network of specialists, partners, and industry contacts that we can actively bring to bear on projects.
Many of our clients appreciate exactly that:
They’re not just working with a design agency—they’re working with a development partner who understands the medtech ecosystem.

When a great idea needs to become a medical device
The situation is quite different when it comes to startups and founders.
It often starts with a strong idea—sometimes even a technological innovation. At the same time, the regulatory landscape is uncharted territory for many young companies. While established companies usually have their own QA and RA (Quality Assurance/Regulatory Affairs) departments, startups and founders face entirely different challenges.
Suddenly, questions like these come to mind:
- What documentation is actually required?
- How do you properly structure development processes?
- What do Notified Bodies or audits expect?
- When must regulatory requirements be taken into account?
During this phase, a structured development partner can be of enormous help. Our approach provides startups with a clear framework for systematically developing their idea. Above all, our QMS offers young companies one thing: guidance and security.
This means:
- Structured product development—a clear development framework that systematically transforms ideas into a robust product concept
- Development decisions are thoroughly documented
- Design processes are structured in a way that makes them easy to follow
- Navigating the regulatory landscape - regulatory requirements are taken into account early on
- Common pitfalls can be avoided
- Access to years of experience and an extensive network of experts
However, something else is just as important:
Experience and collaboration. Many founders benefit from having a partner by their side who has already supported numerous medtech projects—and who knows where challenges typically arise. This often leads to valuable connections: with experts, specialized partners, or other players in the medtech field.
For startups, this means:
. They gain not only design support—but also a development partner who helps them navigate the regulated medical technology landscape with confidence.
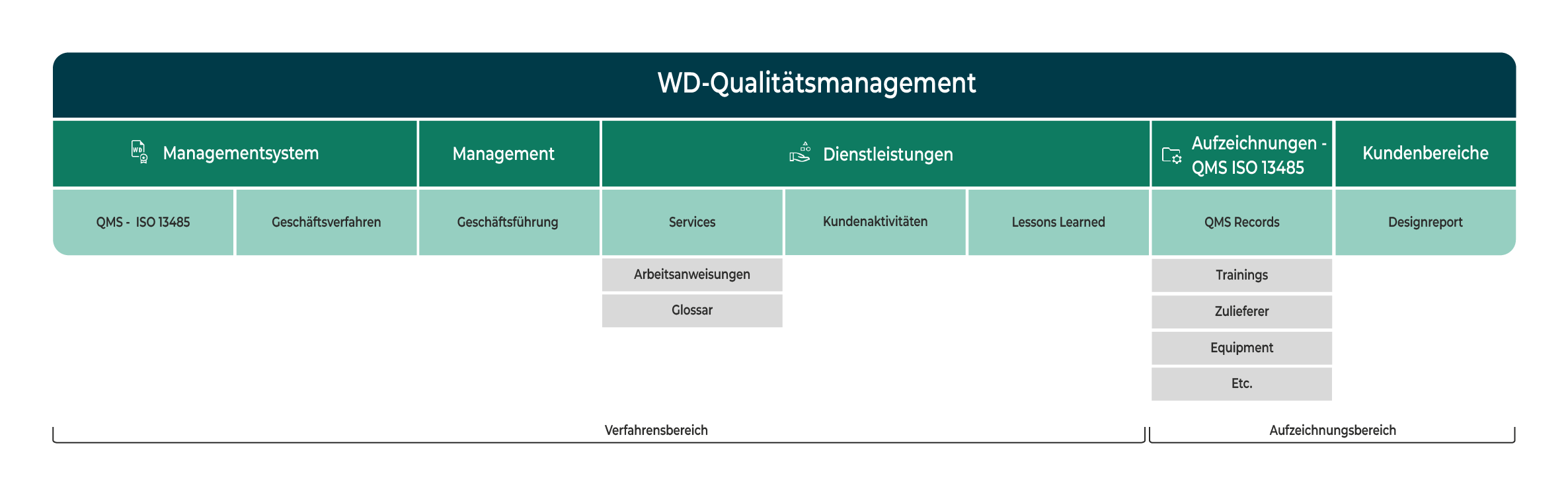
What our WD eQMS means to us:
For this collaboration to work, a clear foundation is needed.
Our quality management system is based on the requirements of ISO 13485, the key quality standard in the medical device industry.
By this we mean:
Our processes are designed to meet the requirements of regulated development projects.
These include, among other things:
- structured design processes
- clear responsibilities
- clear documentation
- Traceability of decisions
- defined interfaces with Quality Assurance (QA) and Regulatory Affairs (RA)
At the same time, we make sure that these structures work in practice and do not become mere bureaucracy. After all, a management system does not derive its value from a certificate—but from the fact that it is actually used in day-to-day operations.
A side effect: better teamwork
A structured framework doesn’t just impact projects with our clients. It also helps internally. Clear processes facilitate collaboration between teams, make knowledge easier to share, and ensure that lessons learned from projects can be systematically built upon.
For our customers, this means one thing above all else:
You benefit from a way of working that remains consistent and adaptable across many projects .
Medical technology is rarely developed in isolation
It arises from the interplay of development, design, regulation, and numerous partners involved. Our approach ensures that we can navigate this environment with confidence.
The result:
Fewer follow-up questions.
Fewer misunderstandings.
Fewer rounds of revisions.
Less rework.
Less stress during the project.
Faster approvals.
Faster and more actionable decisions.
More reliable results.
Greater certainty.
Good medtech design isn’t just functional and user-centered. It’s interoperable, transparent, and compliant with regulatory requirements. And that’s exactly what saves time, money —and stress—in the end .
Frequently asked questions